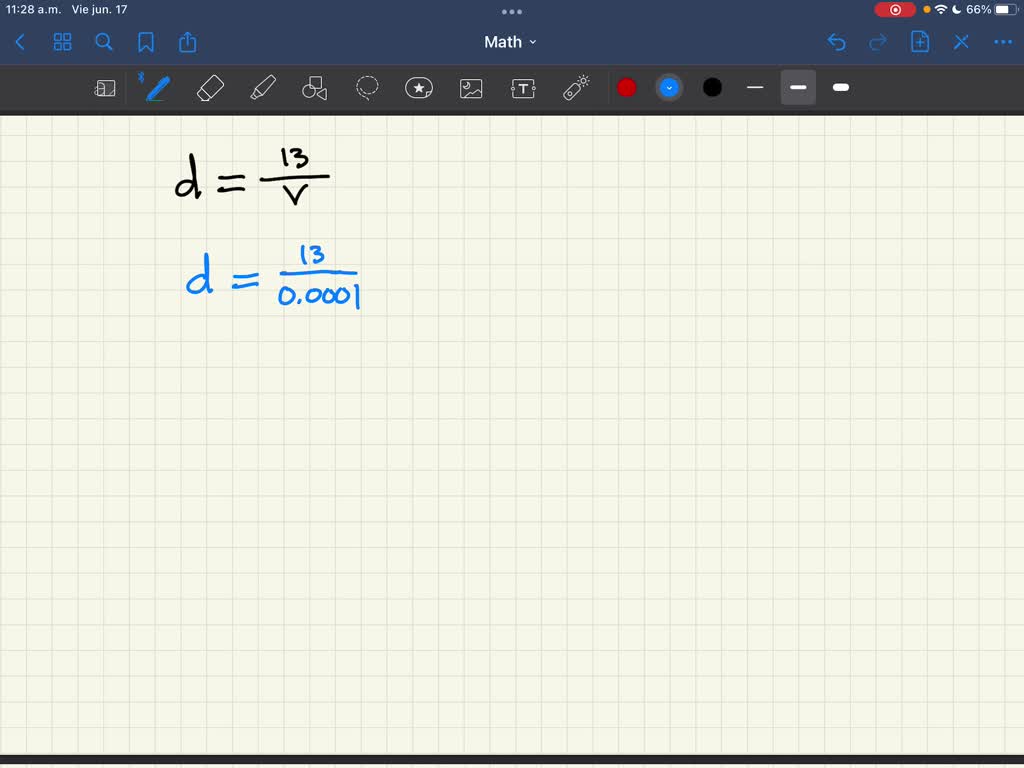
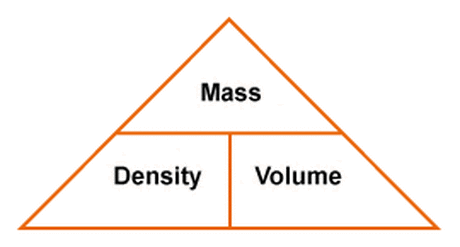
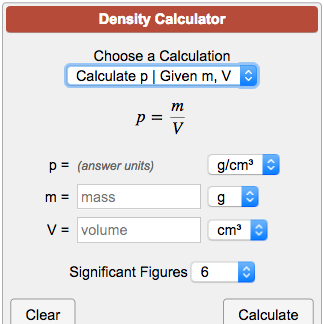
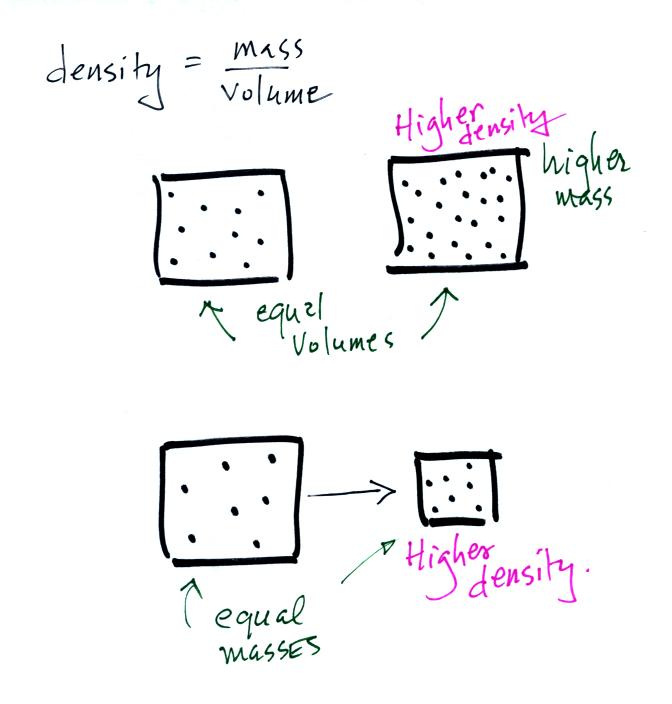
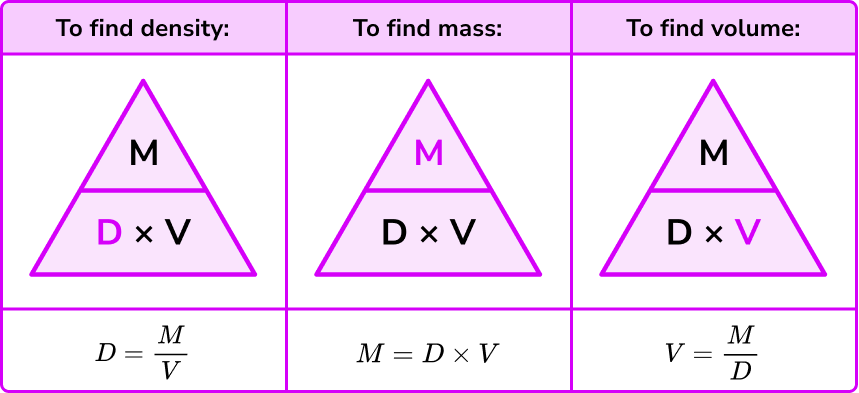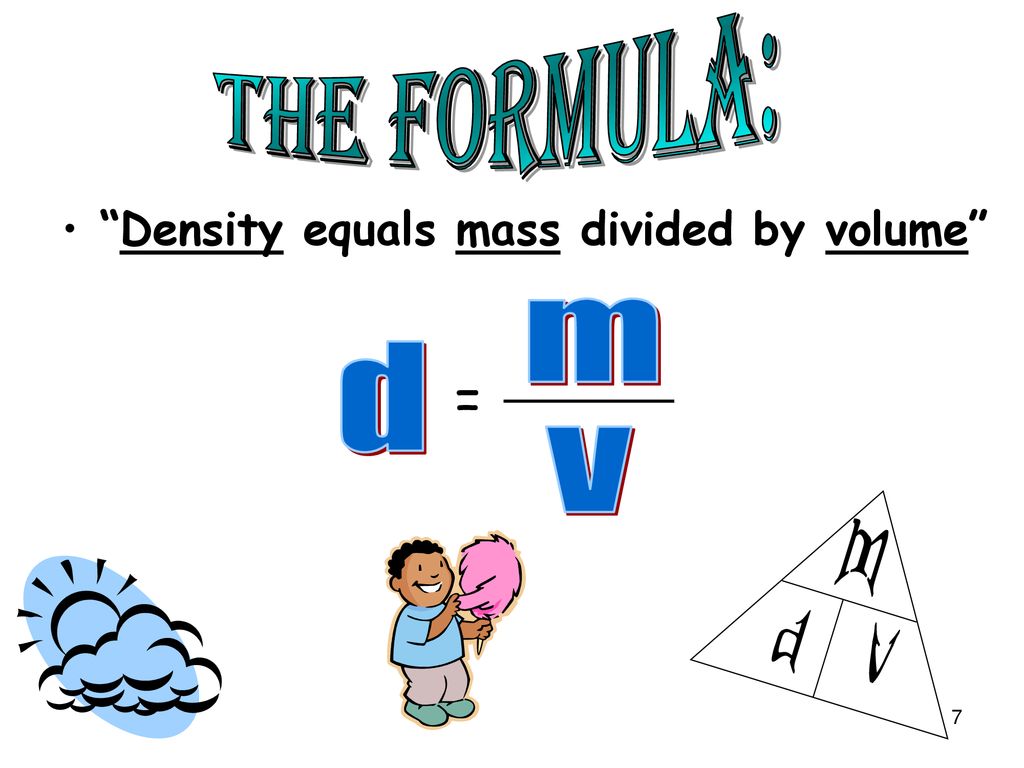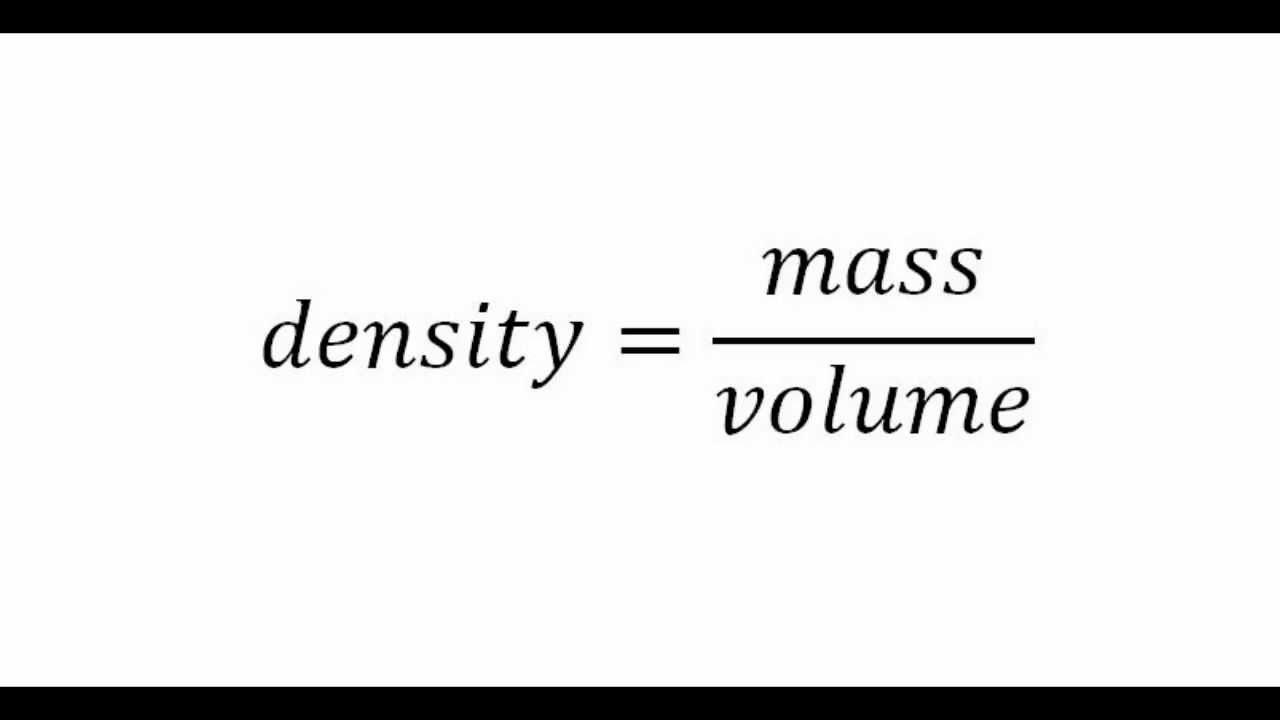Lab/hw) so if the formula is density equals mass over volume, so assume I would subtract mass of empty beaker with mass with unknown liquid and then continue with the formula or

calculus - Could somebody explain why exactly is the density of the lower rod is equal to $f(g(u))$? - Mathematics Stack Exchange

If the density of an object is exactly equal to the density of a fluid, The object will: (a) float - YouTube

SOLVED: The density of a substance equals its mass divided by its volume. Talia listed the density of some common materials at 20 °C. Material / Density (g/cm3) gasoline = 0.70 mercury =